Complete Documentation Checklist for X-Ray Equipment Import in India: AERB, CDSCO & ADC Clearance Guide
Get the complete documentation checklist for importing X-ray equipment in India, including CDSCO MD-15, AERB Type Approval, e-LORA procurement, QA reports, and ADC clearance requirements.
IMPORT/EXPORT & CUSTOMS
3/18/20264 min read


Complete Documentation Checklist for X-Ray Equipment Import in India: A Regulatory Guide to AERB and CDSCO Compliance
The importation of X-ray generating equipment (XGE) into India is a meticulously regulated process designed to balance medical benefits with the inherent health risks of ionizing radiation. This process is governed by two primary authorities: the Atomic Energy Regulatory Board (AERB), which oversees radiological safety under the Atomic Energy Act, 1962, and the Central Drugs Standard Control Organization (CDSCO), which regulates device quality.
Since January 1, 2021, all X-ray machines have been classified as Class C (Moderate to High Risk) medical devices, making mandatory registration a prerequisite for any business plan involving manufacture or import. Failure to adhere to this dual-track compliance leads to significant "supply chain disruption," financial overheads, and customs delays. This guide provides a comprehensive documentation checklist to ensure successful Assistant Drug Controller (ADC) clearance and legal operation.
1. CDSCO Regulatory Prerequisites: The Pathway to MD-15
The first step in the Step-by-Step import process is satisfying the CDSCO requirements under the Medical Device Rules, 2017. X-ray equipment is now considered a "drug" for regulatory purposes, requiring a formal license to enter the Indian market.
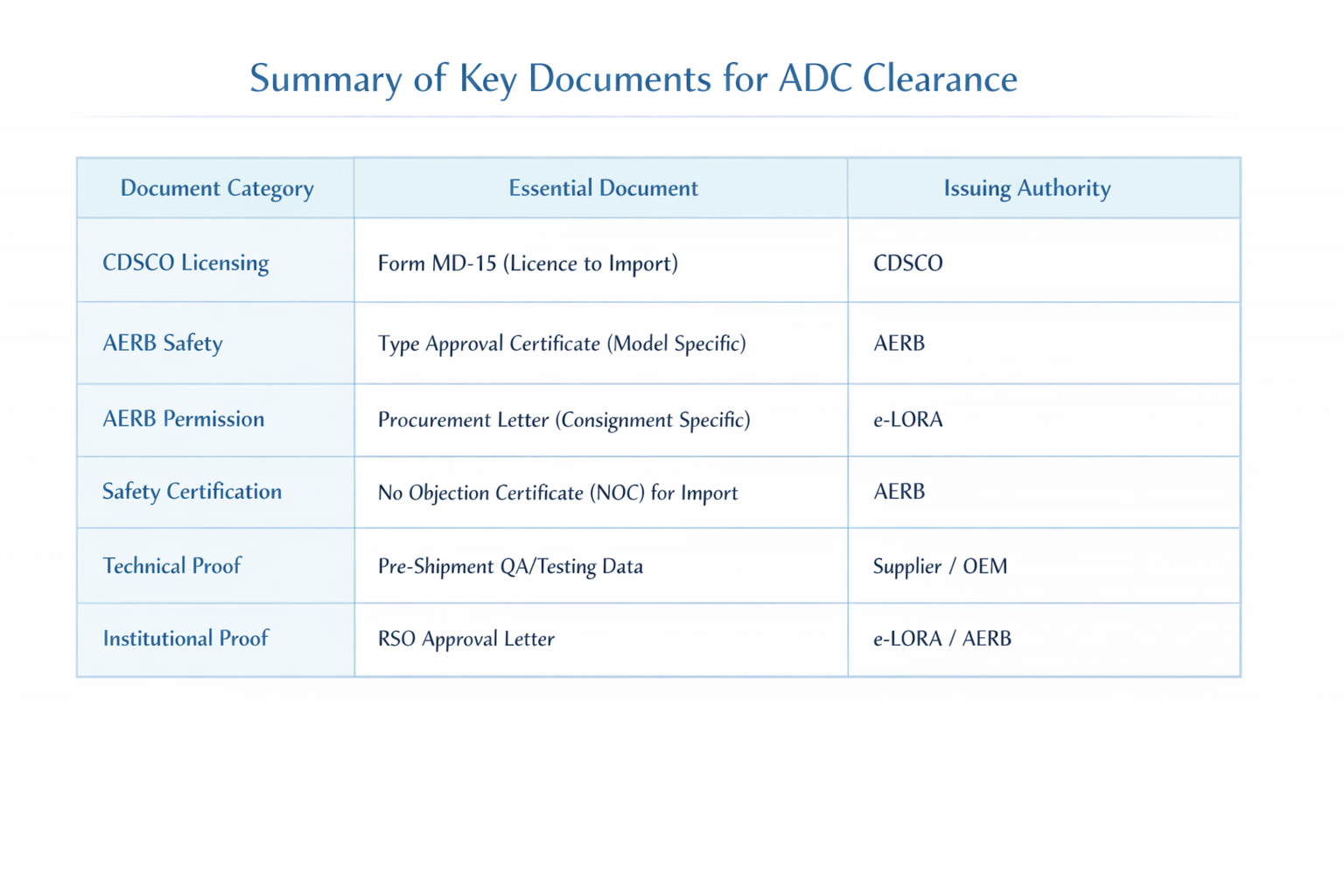
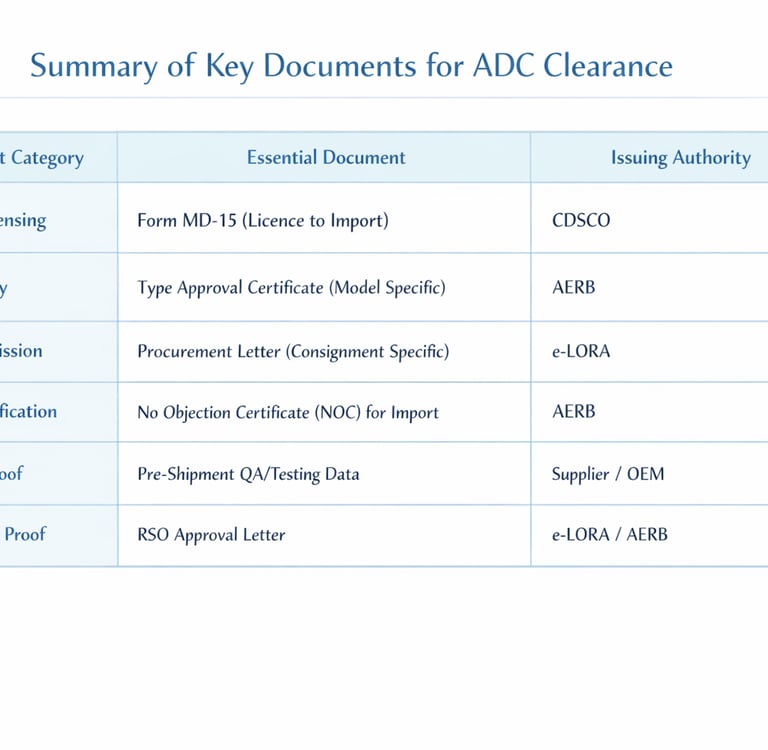
Form MD-14 (Import Licence Application): Importers must submit this application to the Central Licensing Authority to seek an import license for Class C medical devices.
Form MD-15 (Grant of Licence to Import): This is the official document issued by the CDSCO after processing the MD-14. It is mandatory for ADC clearance at the port of entry.
Manufacturing Plant Registration: Documentation must include the registration of the foreign manufacturing site. The government fee for this is currently $3,000 per plant.
Product Registration Fees: A fee of $1,500 per product is required for registration under the Class C category.
Authenticated Regulatory Dossier: This includes manufacturing site details, device performance summaries, and valid international certifications such as CE or ISO.
2. AERB Consenting Framework: Type Approval and NOCs
Parallel to CDSCO registration, every imported model must conform to Indian safety standards as defined in the AERB Safety Code (AERB/RF-MED/SC-3 Rev. 2).
AERB Type Approval Certificate: This is a prerequisite for commercial marketing. It is issued after a technical evaluation of a prototype to ensure the model conforms to national safety standards. Note that Type Approval becomes invalid if design specifications are altered.
No Objection Certificate (NOC) for Import: If a supplier intends to market a foreign model for the first time, they must obtain an NOC from the AERB specifically for the purpose of demonstrating performance for Type Approval.
Permission for Procurement: Once a model is Type Approved, the importer must obtain a specific permission for procurement for each individual consignment through the e-LORA portal.
3. Institutional Readiness: The e-LORA Portal Documentation
The e-LORA (e-Licensing of Radiation Applications) portal is the mandatory digital gateway for all regulatory transactions in India. Before equipment arrives, the facility (the Employer) must be digitally prepared.
Institute Registration Documents: The facility must register on e-LORA using valid login details, a registered email, and a mobile number.
RSO Designation and Approval: Every facility must have a Radiological Safety Officer (RSO) approved by the Competent Authority. For high-risk facilities like CT and Interventional Radiology, the RSO must be a radiologist or technologist with at least three years of field experience.
Personnel Monitoring Tie-up: Documentation proof of an agreement with a BARC-approved TLD (Thermoluminescent Dosimeter) agency is required.
Radiation Protection Programme (RPP): A written plan for controlling, monitoring, and assessing occupational and public exposures must be maintained by the licensee.
4. Mandatory Technical Dossier and Built-in Safety Benchmarks
For customs clearance and subsequent licensing for operation, the equipment must meet strict technical benchmarks. These specifications should be detailed in the supplier's instruction manual and technical data sheets.
X-Ray Tube Housing Integrity: Documentation must confirm that radiation leakage does not exceed 1 mGy in one hour at 1 meter for radiography units, or 0.25 mGy/hr for dental intra-oral (IOPA) units.
Beam Filtration Specifications: The total filtration must be at least 2.5 mm Al equivalent for equipment operating at constant potential to protect patients from "soft" X-rays.
Cable Length Verification: For mobile and portable radiography units, documentation must state that the exposure cable length is not less than 2 meters.
Weight Limit for Portables: Equipment intended for field use must have a documented weight not exceeding 12 kg to be classified as "portable".
Warning Signage and Marking: The X-ray tube housing must be conspicuously marked with the make, model, serial number, focal spot position, and the equilateral triangle radiation caution symbol.
5. Post-Import Installation and Commissioning Documents
Clearing the ADC is only one phase; the equipment cannot be legally used for patient diagnosis until it is "commissioned" and licensed for operation.
Installation Report: After delivery, an authorized engineer must install the unit and generate an installation report, which is then uploaded to e-LORA.
Quality Assurance (QA) Report: The importer must ensure a Third-Party QA check is conducted by an authorized agency. This report verifies critical parameters like kVp accuracy (within ± 5 kV) and timer accuracy (error ≤ ± 10%).
Acceptance Testing Documentation: This confirms that the functional performance of the unit matches the baseline values provided by the manufacturer.
Instruction Manuals: The supplier must provide manuals in English/Hindi covering safe operation, periodic inspection, and emergency procedures.
6. Administrative Compliance Checklist for Facilities
The Employer acts as the custodian of the equipment and holds ultimate responsibility for safety.
Logbook Maintenance: A logbook must be maintained to record the use and operation of the unit, including daily storage times for portable field units.
Service and Maintenance Records: Periodic servicing must be carried out by authorized agencies, and records of any component replacements (like X-ray tube inserts) must be kept for regulatory review.
Occupational Exposure Records: Annual dose records for every radiation worker must be furnished to the staff and made available for AERB inspections.
Insurance and Warranty Documentation: For public health implementations (such as TB screening), devices must be insured and under warranty for at least the first two years.
Summary of Key Documents for ADC Clearance
Conclusion: Upholding Expertise and Trust in Radiology
Adhering to this complete documentation checklist is the cornerstone of Expertise, Authoritativeness, and Trustworthiness (E-E-A-T) in India's radiology sector. By ensuring that every X-ray unit—from ultra-portable handhelds to high-end CT scanners—is backed by MD-15 licenses, AERB Type Approvals, and rigorous Quality Assurance, importers and clinicians fulfill their duty of care.
Compliance ensures that diagnostic information is obtained at the lowest possible radiation risk to the patient, adhering to the international ALARA (As Low As Reasonably Achievable) principle. Ultimately, a well-documented installation is not only a legal requirement under the Atomic Energy Act but a commitment to public health and professional excellence.
About XRAYNEWS.NET
XRAYNEWS.NET provides updates on medical and industrial X-ray technologies, radiography insights, regulatory awareness, and imaging industry developments.
Regulatory Resources
© 2025. All rights reserved.
Video Resources
Popular Topics
Quick Links
Stay Updated with X-Ray Industry News
Subscribe to receive updates on X-ray technology developments, radiography insights, and industry news.
