OEM & SKD Export Model in X-Ray Industry: Compliance Guide for Indian Manufacturers
Learn how OEM and SKD export models work in the X-ray industry. Complete guide covering CDSCO, AERB compliance, QA standards, and global market strategy for Indian manufacturers.
IMPORT/EXPORT & CUSTOMS
4/16/20264 min read

OEM & SKD Export Model in X-Ray Industry: A Strategic Guide for Indian Manufacturers
The Indian diagnostic imaging sector is undergoing a massive transformation, shifting from a domestic focus to becoming a global hub for X-ray generating equipment (XGE). This evolution is governed by a strict dual-regulatory framework led by the Atomic Energy Regulatory Board (AERB) and the Central Drugs Standard Control Organization (CDSCO). For Indian manufacturers, adopting an Original Equipment Manufacturer (OEM) or Semi-Knocked Down (SKD) export model requires deep expertise in radiation safety, "built-in" design safety, and international quality benchmarks. This guide provides an authoritative roadmap for manufacturers to navigate certifications, technical standards, and global compliance for exporting X-ray technology.
1. Defining the OEM and Manufacturing Framework
In the Indian regulatory landscape, the Original Equipment Manufacturer (OEM) is defined as any person or entity involved in the manufacturing of X-ray equipment and X-ray tubes. The legal framework for this is established by the Atomic Energy (Radiation Protection) Rules, 2004, and the Atomic Energy Act, 1962.
Ultimate Responsibility: The prime responsibility for ensuring radiation safety in handling an X-ray unit rests with the Employer (Licensee), who acts as the custodian of the equipment.
Outsourced Activity: If an OEM chooses to outsource part or all of its manufacturing (an essential component of the SKD model), the OEM remains responsible for ensuring that all quality protocols regarding radiation safety are strictly followed by the outsourced agency.
Type Approval Prerequisite: Every model of X-ray equipment must obtain a Type Approval Certificate from the Competent Authority before it can be manufactured or supplied on a commercial scale.
2. CDSCO Regulatory Compliance: The Class C Mandate
A fundamental shift occurred on January 1, 2021, when all X-ray machines in India were officially reclassified as Class C (Moderate to High Risk) medical devices. This classification treats X-ray machines as "drugs" for regulatory purposes, necessitating mandatory registration before any commercial movement.
A. Mandatory Licencing Forms and Fees
Manufacturers intending to sell or export must secure the following from the CDSCO:
Form MD-7: The application for a license to manufacture Class C medical devices.
Form MD-9: The official license granted to manufacture for sale or distribution.
Fee Structure: The government fee for manufacturing plant registration is approximately ₹50,000, with an additional ₹1,000 per product registered.
Risk Mitigation: CDSCO registration is required to prevent the proliferation of misbranded or inferior products that could lead to clinical misdiagnosis.
3. AERB Consenting Process and e-LORA Workflow
The e-LORA (e-Licensing of Radiation Applications) portal is the mandatory digital interface for managing the entire lifecycle of an X-ray unit.
Institute Registration: The manufacturing facility must first register as an "Institute" on the e-LORA server.
Procurement Permission: Manufacturers must obtain permission for the procurement or import of X-ray tubes or tube inserts before beginning production.
Type Approval Maintenance: Type Approval is issued based on the demonstration of performance by a prototype; this certificate becomes invalid if any change is made to the design specifications of the approved model.
Decommissioning Support: OEMs are legally responsible for providing technical support to end-users for the decommissioning of equipment once its useful life is over.
4. Technical Excellence: High-Frequency and Design Safety
To compete in global markets, Indian manufacturers must prioritize High-Frequency (HF) technology and superior imaging components.
A. High-Frequency Constant Potential Generators
Modern systems utilize high-frequency constant potential waveforms, which are far more efficient than conventional technology.
Dose Optimization: Constant potential generators (e.g., 60-70 kVp) eliminate "soft" low-energy X-rays that increase patient dose without aiding the image.
Total Filtration: AERB mandates a minimum total filtration of 2.5 mm Al equivalent for constant potential equipment to cut off useless radiation.
B. Focal Spot and Image Resolution
The effective focal spot size—the projected length and width of the spot on the anode—is the primary determinant of image detail.
Handheld Superiority: Many modern handheld OEMs utilize a small 0.4 mm focal spot, which research shows results in significantly higher Line Pair (LP) resolution (6.05–6.55 lp/mm) compared to the 0.7 mm spots found in many traditional wall-mounted units.
5. Safety Infrastructure for Manufacturing Facilities
Manufacturers must maintain a rigorous Radiation Protection Programme (RPP) within their own facilities.
Radiological Safety Officer (RSO): Every facility must designate an AERB-approved RSO who holds a basic degree in Science or a Diploma in Engineering and has completed a prescribed RSO certification course.
Personnel Monitoring (TLD): All workers involved in manufacturing, testing, or servicing must use Thermoluminescent Dosimeter (TLD) badges. These badges must be worn below the lead apron and exchanged quarterly for dose reporting.
Leakage Limits: The X-ray tube housing must be so constructed that leakage radiation does not exceed 1 mGy in one hour at 1 meter for radiography units. For specialized dental units, the limit is stricter at 0.25 mGy/hour.
Quality Assurance (QA) Equipment: The facility must possess calibrated QA equipment traceable to national or international standards to verify parameters like kVp accuracy (± 5 kV) and timer accuracy (± 10%).
6. The Rising Global Demand: Ultra-Portable Systems
One of the "hottest" export opportunities for Indian OEMs lies in ultra-portable (UP) and handheld systems, particularly for the National Tuberculosis Elimination Programme and similar global health initiatives.
Portability Definition: AERB defines portable units as equipment weighing no more than 12 kg that can be carried by one or two persons.
Field Utility: These battery-powered devices pair with Digital Radiography (DR) detectors, often using highly sensitive LG Oxide technology (e.g., 14" x 17" panels).
AI Integration: UP systems are frequently paired with Artificial Intelligence (AI) and Computer-Aided Detection (CAD) software for automated, standardized interpretation of Chest X-rays (CXR).
Field Safety: Portable scanners used in the field must allow for remote operation from a distance of at least 30 meters to ensure the public dose does not exceed 5 µSv per scan.
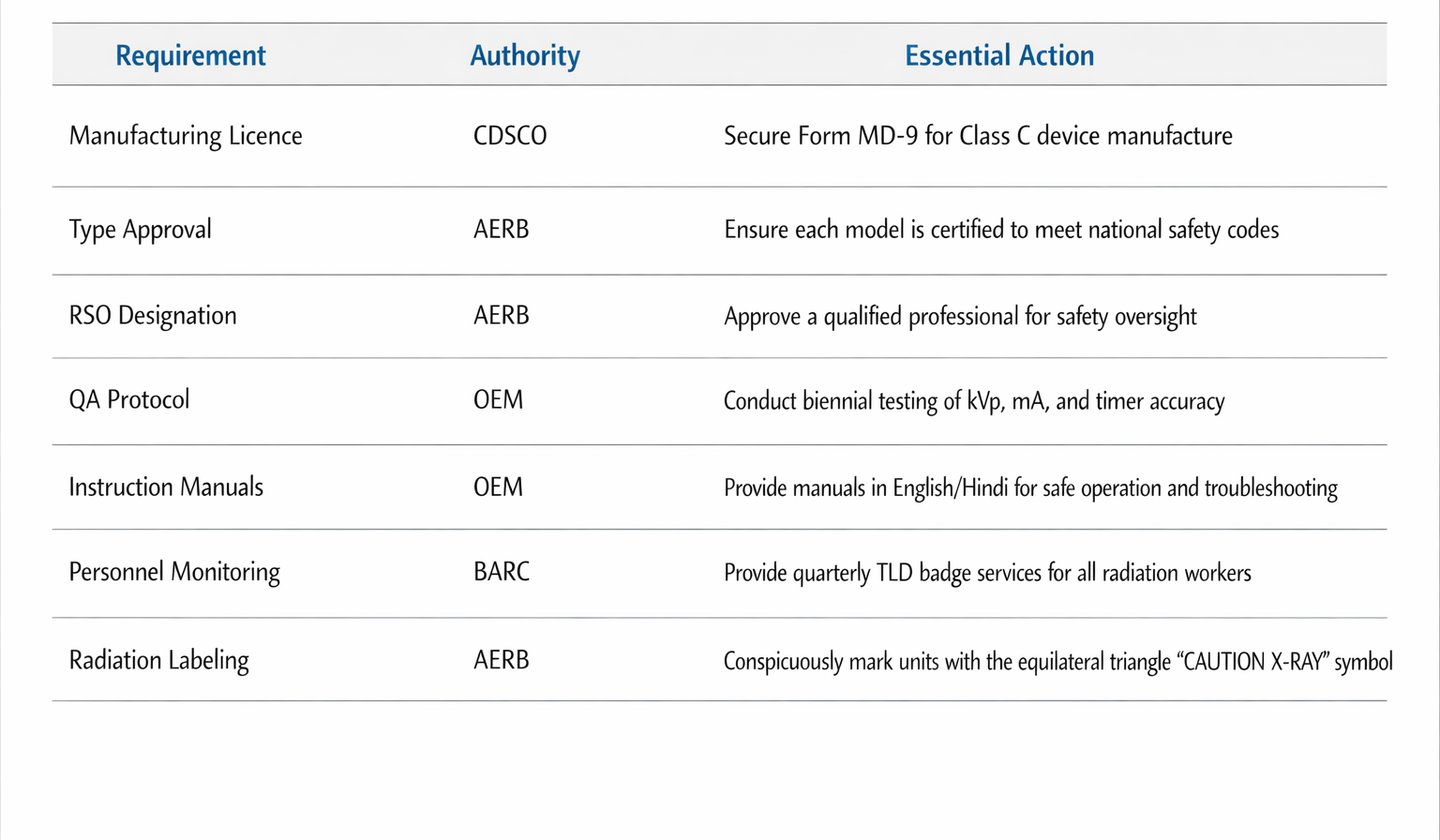
7. Summary Checklist for Export Success
Indian manufacturers aiming for a successful OEM/SKD export model should adhere to the following checklist:
Conclusion: Upholding Expertise and Global Trust
Adopting an OEM or SKD model in the X-ray industry is a commitment to Expertise, Authoritativeness, and Trustworthiness (E-E-A-T). By meticulously following the AERB and CDSCO regulatory pathways, Indian manufacturers can ensure that their products fulfill the ALARA (As Low As Reasonably Achievable) principle—providing the highest quality diagnostic information at the lowest possible radiation risk. Success in the global market is not just about technical specs; it is about a robust culture of safety, documented Quality Assurance, and a religious adherence to national and international standards. As technology miniaturizes and AI becomes more integrated, the Indian X-ray industry is poised to lead the global supply chain for safe, high-resolution diagnostic tools.
About XRAYNEWS.NET
XRAYNEWS.NET provides updates on medical and industrial X-ray technologies, radiography insights, regulatory awareness, and imaging industry developments.
Regulatory Resources
© 2025. All rights reserved.
Video Resources
Popular Topics
Quick Links
Stay Updated with X-Ray Industry News
Subscribe to receive updates on X-ray technology developments, radiography insights, and industry news.
