How to Export X-Ray Machines from India: Certifications, Compliance & Global Market Entry
Learn how to export X-ray machines from India with CDSCO, AERB, and international certifications. Complete guide to compliance, QA standards, and global market entry.
IMPORT/EXPORT & CUSTOMS
4/15/20264 min read


How to Export X-Ray Machines from India: Certifications, Compliance & Global Market Entry
The Indian diagnostic imaging industry has matured into a high-technology sector, making India an emerging hub for the export of X-ray generating equipment (XGE). Under the Atomic Energy Act, 1962, and the Medical Device Rules, 2017, the Indian government has established a world-class regulatory environment that ensures "built-in safety" and functional excellence. For manufacturers aiming at global market entry, "handling" X-ray equipment—which legally includes the process of export—requires strict adherence to the protocols of the Atomic Energy Regulatory Board (AERB) and the Central Drugs Standard Control Organization (CDSCO). This guide provides a strategic overview of the certifications, technical benchmarks, and compliance steps necessary to successfully export "Made in India" X-ray systems.
1. Navigating the Regulatory Framework for Exports
In India, the radiological safety of X-ray equipment is governed by the Atomic Energy (Radiation Protection) Rules, 2004, while device quality is regulated by the CDSCO.
The Competent Authority: The Chairman of the AERB is the Competent Authority responsible for enforcing radiation safety standards across the equipment's life cycle, including its manufacture and transfer for export.
Medical Device Classification: Since January 1, 2021, all X-ray machines are classified as Class C (Moderate to High Risk) medical devices. This means they are legally considered "drugs" for regulatory purposes, necessitating a formal manufacturing license before they can be sold or exported.
Mandatory Registration: No manufacturer can place X-ray equipment in the market (domestic or international) without mandatory registration with the CDSCO to prevent the proliferation of inferior-quality products that lead to misdiagnosis.
2. Essential Certifications for Global Market Entry
To compete in the international market, Indian manufacturers must possess a suite of regulatory consents that prove their equipment meets both national and international safety criteria.
A. CDSCO Manufacturing Licences
Manufacturers must apply for the grant of a license to manufacture for sale or distribution using specific forms:
Form MD-7: The application for the issue of a manufacturing license for Class C medical devices.
Form MD-9: The official license granted by the CDSCO for manufacturing Class C devices.
Fees: Government fees are approximately ₹50,000 for plant registration and ₹1,000 per product.
B. AERB Type Approval
Every model intended for export must first obtain an AERB Type Approval Certificate.
Prototype Testing: Approval is issued only after a prototype model demonstrates satisfactory performance in a dedicated radiation testing facility.
Validity: Type Approval verifies that the device conforms to the AERB Safety Code and remains valid as long as no changes are made to the design specifications.
C. International Standards Conformity
For imported components or for export to regulated markets, the equipment must meet international standards such as IEC, CE, or ISO. The AERB requires authenticated reports confirming that the design meets these benchmarks.
3. Technical Benchmarks: Meeting International Quality Standards
Global buyers, particularly in the public health sector, demand equipment that optimizes image quality while minimizing radiation dose.
Generator Technology: High-frequency (HF) constant potential generators are the global gold standard. They eliminate low-energy "soft" X-rays that increase patient dose without aiding the diagnostic image.
Radiation Leakage Limits: For medical radiography, leakage through the protective tube housing must not exceed 1 mGy in one hour at 1.0 meter. For dental intra-oral (IOPA) units, the limit is stricter at 0.25 mGy/hr.
Beam Filtration: Systems operating at constant potential must have a minimum total filtration of 2.5 mm Al equivalent to ensure radiation quality.
Focal Spot Size: Smaller focal spots (e.g., 0.4 mm) are preferred for high Line Pair (LP) resolution, allowing for the visualization of minute anatomical details.
4. Institutional Compliance: The e-LORA and RSO Mandate
The e-LORA (e-Licensing of Radiation Applications) portal is the mandatory digital gateway for managing the regulatory lifecycle of X-ray equipment in India.
Institute Registration: The manufacturing facility must be registered as an institute on the e-LORA server with valid credentials.
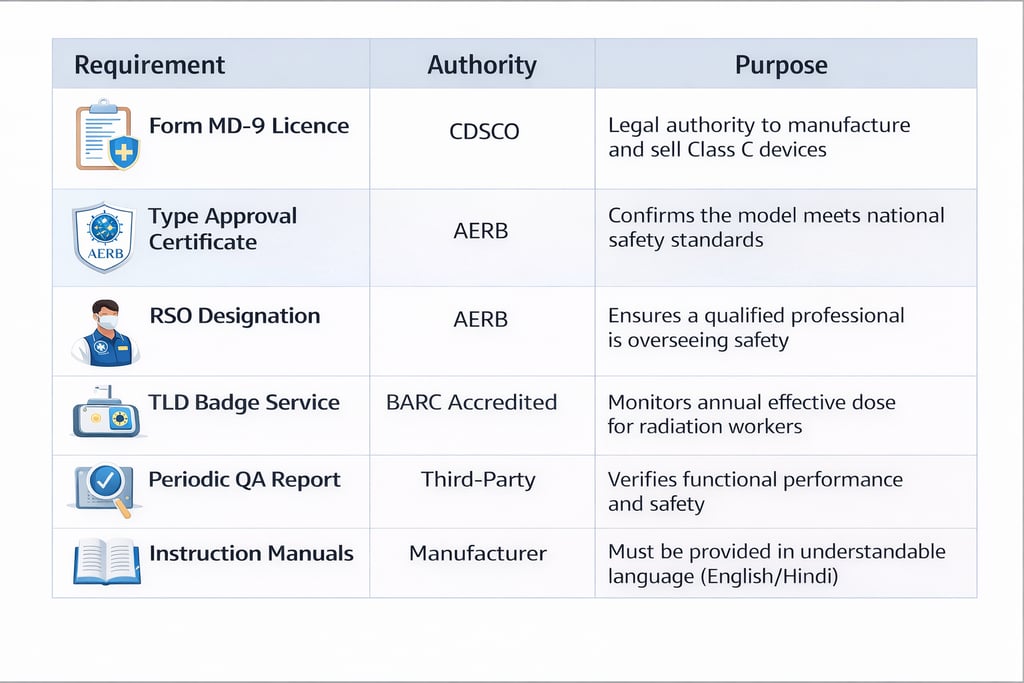
Radiological Safety Officer (RSO): Every manufacturing facility is legally required to designate an AERB-approved RSO.
Qualifications: The RSO must have a basic degree in Science or a Diploma in Engineering and have completed an AERB-prescribed certification course.
Responsibilities: The RSO must implement a Radiation Protection Programme (RPP), maintain Quality Assurance (QA) records, and monitor occupational doses for all radiation workers using TLD badges.
5. The "Hot" Export Market: Ultra-Portable AI-Integrated Systems
India's success in global markets is increasingly tied to the rise of ultra-portable (UP) and handheld X-ray systems, which are ideal for community-based screening in low-resource settings.
Portability Standards: AERB defines portable equipment as units intended to be carried by one or two persons, with a weight not exceeding 12 kg.
Public Health Application: These systems are in high demand for Tuberculosis (TB) screening programs, particularly when paired with Artificial Intelligence (AI) and Computer-Aided Detection (CAD) software.
CAD Advantage: AI-powered CAD tools provide automated, standardized interpretation of Chest X-rays (CXR), which is vital in regions where expert human readers are scarce.
Field Safety: Portable scanners used in the field must allow for remote operation from a distance of at least 30 meters to keep public doses below 5 µSv per scan.
6. Steps to Ensure Seamless Export Clearance
Manufacturers must follow a meticulous "Step-by-Step" process to avoid supply chain disruptions or customs delays.
Grant of CDSCO MD-9 Licence: Ensure the manufacturing site and specific products are registered under the Medical Device Rules.
Type Approval Maintenance: Verify that each model's Type Approval is current and that no design changes have invalidated the certification.
Quality Assurance (QA) Certification: Conduct periodic QA tests (every two years or after major repairs) through authorized agencies to verify parameters like kVp accuracy (± 5 kV) and timer accuracy (± 10%).
Authorized Supplier Network: Only AERB-authorized suppliers or the Original Equipment Manufacturer (OEM) can legally transfer or sell XGE for export.
Documentation Dossier: Maintain an exhaustive record of testing data, RSO approvals, and personnel monitoring (TLD) reports for regulatory review.
7. Summary Checklist for Export Readiness
Conclusion
Exporting X-ray machines from India is a prestigious endeavor that demands a deep commitment to Expertise, Authoritativeness, and Trustworthiness (E-E-A-T). By aligning with the rigorous standards of the AERB and CDSCO, Indian manufacturers can offer high-quality, "safe-by-design" products to the global market. Success depends on maintaining a robust culture of safety—centered on the ALARA (As Low As Reasonably Achievable) principle—and utilizing digital gateways like e-LORA to manage compliance. As technology continues to miniaturize and integrate with AI, India is well-positioned to lead the global supply of portable diagnostic tools, provided every export is backed by rigorous Quality Assurance and a valid regulatory license.
About XRAYNEWS.NET
XRAYNEWS.NET provides updates on medical and industrial X-ray technologies, radiography insights, regulatory awareness, and imaging industry developments.
Regulatory Resources
© 2025. All rights reserved.
Video Resources
Popular Topics
Quick Links
Stay Updated with X-Ray Industry News
Subscribe to receive updates on X-ray technology developments, radiography insights, and industry news.
