How to Avoid Customs Delays in X-Ray Equipment Import | CDSCO & AERB Guide India
Avoid customs delays in X-ray equipment import in India. Learn CDSCO MD-15, AERB approval, e-LORA process, and compliance checklist for smooth clearance.
IMPORT/EXPORT & CUSTOMS
4/17/20265 min read


How to Avoid Customs Delays in X-Ray Equipment Import: Expert Tips
The diagnostic imaging landscape in India is currently defined by a rigorous dual-regulatory framework intended to eliminate the risks associated with misbranded, inferior-quality products that lead to misdiagnosis and inaccurate results. Since January 1, 2021, X-ray machines have been classified as Class C (Moderate to High Risk) medical devices, making mandatory registration a legal prerequisite for import. Navigating the requirements of the Central Drugs Standard Control Organization (CDSCO) and the Atomic Energy Regulatory Board (AERB) is essential for a smooth Assistant Drug Controller (ADC) clearance. Failure to manage these regulatory documents can lead to complete delays in business plans, excessive overheads, and severe supply chain disruption.
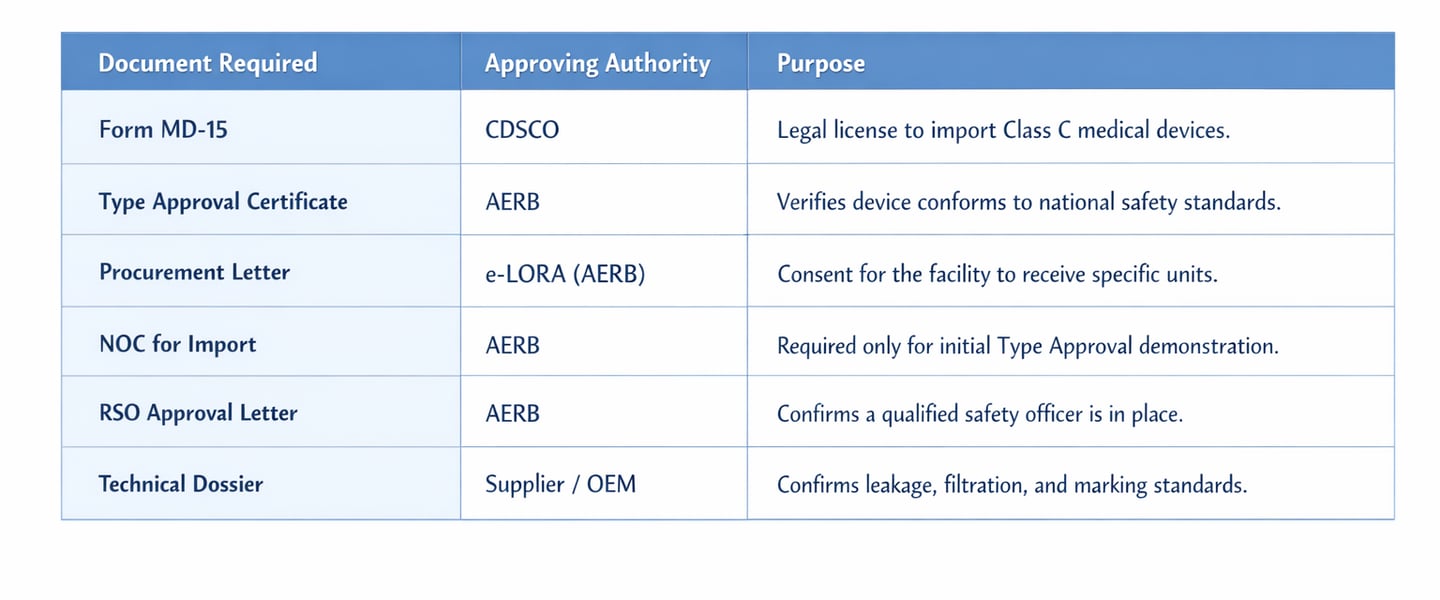
1. Secure Your CDSCO MD-15 Import Licence Early
Under the Medical Device Rules 2017, X-ray Generating Equipment (XGE) is legally considered a "drug" for regulatory purposes. To legally import these devices, you must obtain a formal license from the CDSCO, the Central Licensing Authority.
Application Process (Form MD-14): Importers must submit an application through Form MD-14 to seek an import license for Class C or D devices.
The Grant of Licence (Form MD-15): The official MD-15 license is the critical document required by customs to verify that the equipment meets national quality standards.
Government Fees: Budgeting for these fees is a vital part of financial planning. Currently, registration costs $3,000 for manufacturing plant registration and $1,500 per individual product.
Preventing Misdiagnosis: This gatekeeping mechanism was specifically implemented to curb a spurt in poor-quality devices that caused serious implications for patients.
2. Master the AERB Consenting Framework
While the CDSCO handles device quality, the AERB ensures radiological safety under the Atomic Energy (Radiation Protection) Rules, 2004. The AERB mission is to ensure that ionizing radiation use does not cause undue risk to human health or the environment.
The Type Approval Prerequisite
No X-ray equipment can be marketed or operated in India unless it is Type Approved by the Competent Authority (Chairman, AERB).
Performance Demonstration: Type Approval is issued based on the evaluation of a prototype to ensure it conforms to safety standards.
Validity: This certificate becomes invalid if any design specifications of the model are changed.
New Foreign Models: If a model is not yet Type Approved in India, the authorized supplier must first obtain a No Objection Certificate (NOC) for Import specifically for demonstration purposes.
Konsignment-Wise Procurement Permission
Even for Type Approved models, routine import requires an equipment-specific Permission for Procurement for every single consignment. This is handled digitally through the e-LORA (e-Licensing of Radiation Applications) portal.
3. Institutional Readiness and e-LORA Compliance
A frequent cause of customs bottlenecks is the lack of institutional registration on the e-LORA server. The Employer (the person having overall responsibility for the facility) must ensure the institute is registered before procurement.
RSO Designation: Every facility must have a Radiological Safety Officer (RSO) approved by the Competent Authority. For high-risk facilities like CT and Interventional Radiology, the RSO must be a radiologist or a technologist with at least three years of working experience in the field.
Pending Tasks: Before applying for a new procurement letter, ensure there are no pending notifications for the facility (e.g., overdue radiation checks or training) as this can delay the approval of new applications.
The Procurement Letter Workflow: Select the "Equipment manufactured in abroad" tab in e-LORA and ensure the model name matches the manufacturer's serial number and technical dossier exactly.
4. Technical Built-in Safety Benchmarks
Customs officials and ADC inspectors often verify the "built-in safety" of the equipment against the AERB Safety Code (SC-3).
Radiation Leakage Limits: For general radiography, leakage through the protective tube housing must not exceed 1 mGy in one hour at 1.0 meter from the focus. For dental intra-oral (IOPA) units, the limit is stricter at 0.25 mGy/hour.
Beam Filtration: Equipment operating at constant potential must have a minimum total filtration of 2.5 mm Al equivalent to absorb low-energy components that do not contribute to diagnostic images.
Cable Length for Portables: Mobile and portable X-ray equipment must be provided with an exposure cable length of not less than 2 meters to ensure the operator can maintain a safe distance during activation.
Mandatory Identification: The X-ray tube housing must be conspicuously marked with the make, model, serial number, focal spot position, and inherent/added filtration.
5. Safety Infrastructure and Personnel Monitoring
Your facility must demonstrate that it can handle the equipment safely before it clears customs and is licensed for operation.
Personnel Monitoring (TLD Badges): All radiation workers handling the equipment must be provided with Thermoluminescent Dosimeter (TLD) badges from BARC-accredited laboratories.
Lead Equivalence Requirements: You must procure mandatory radiation protection devices, including:
Protective Aprons: Minimum 0.25 mm lead equivalence.
Mobile Protective Barriers: Minimum 1.5 mm lead equivalence with a viewing window.
Shielded Doors: X-ray room doors require a lead lining of 1.7 mm.
Warning Signage: An equilateral triangle radiation symbol with "CAUTION-X-RAY" in English, Hindi, and the local language must be ready for posting on the entrance door.
6. Step-by-Step Logistics Checklist
To minimize human error and administrative gaps, follow this authoritative sequence for import:
Grant of MD-15: Ensure your CDSCO registration is active for the manufacturing plant and the specific model.
Type Approval Verification: Confirm the model has a valid Type Approval Certificate from AERB.
e-LORA Procurement Letter: Apply for and download the Permission for Procurement letter. This usually takes only a few hours from submission if all data is correct.
Authorized Supplier Check: Ensure the unit is supplied by an authorized person recognized by the manufacturer and AERB.
Pre-Shipment QA: Verify that the manufacturer has conducted Acceptance Testing/Quality Assurance checks to confirm parameters like kVp accuracy (± 5 kV) and timer accuracy (± 10%) meet Indian standards.
Transportation Integrity: Equipment like ultraportable units (weight < 12 kg) must be transported in a designated case with a number-coded lock and stored in a sanitary environment free from dust and water leakages.
7. Summary Table: Essential Documents for Customs Clearance
Conclusion: Upholding Expertise and Trustworthiness
Achieving diagnostic excellence requires more than just high-tech hardware; it demands a deep commitment to Expertise, Authoritativeness, and Trustworthiness (E-E-A-T) by adhering to the rigorous safety protocols established by the government. By following the ALARA (As Low As Reasonably Achievable) principle and ensuring all administrative, technical, and personnel requirements are met well in advance, importers can avoid the legal penalties of the Atomic Energy Act, 1962, which include suspension of licenses, fines, or imprisonment. Success in the Indian X-ray market is built on the foundation of religious compliance—ensuring that every digital image is obtained at the lowest possible radiation risk to the patient.


About XRAYNEWS.NET
XRAYNEWS.NET provides updates on medical and industrial X-ray technologies, radiography insights, regulatory awareness, and imaging industry developments.
Regulatory Resources
© 2025. All rights reserved.
Video Resources
Popular Topics
Quick Links
Stay Updated with X-Ray Industry News
Subscribe to receive updates on X-ray technology developments, radiography insights, and industry news.
